Glutamic Acid-Chelated Cobalt Stabilizes G-Quadruplexes and Selectively Suppresses Hepatocellular Carcinoma Growth
GACC reduced tumor burden ≈ 90% in zebrafish xenografts at 50 μM with no detected hepatotoxicity — supports follow-up PK/ADME and mammalian studies.
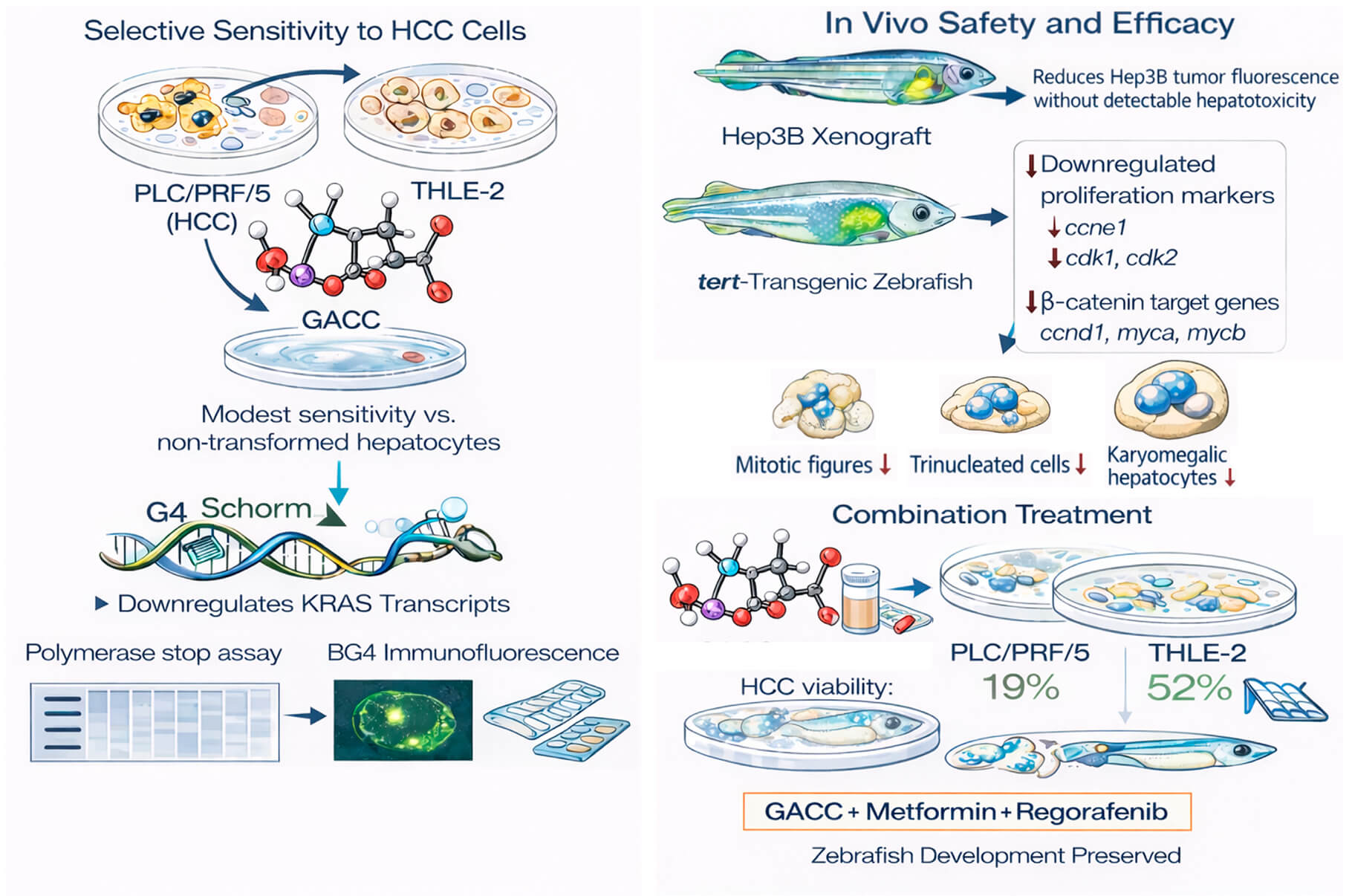
Objectives: Hepatocellular carcinoma (HCC) has limited systemic options with substantial toxicity. G-quadruplex (G4) structures in oncogene promoters are attractive but challenging drug targets. This study aimed to determine whether glutamic acid–chelated cobalt (GACC) is a G4-active scaffold with anti-HCC efficacy and favorable in vivo safety.
Methods: Anticancer activity was tested in HCC cell lines (PLC/PRF/5, Hep3B, HepG2) and non-transformed THLE-2 hepatocytes. In vivo safety/efficacy were assessed in zebrafish embryo toxicity assays, a Hep3B xenograft model, and a tert-overexpressing transgenic zebrafish model.
Results: GACC reduced HCC viability (IC50 ~86–115 µM) and showed low embryotoxicity. In zebrafish xenografts, GACC (50 µM) reduced Hep3B tumor fluorescence by ~90% without detectable hepatotoxicity. GACC suppressed proliferation and reduced KRAS transcripts.
Conclusion: GACC is a G4-active cobalt–glutamate scaffold with anti-HCC activity and favorable zebrafish safety, enabling rational, less toxic combination design.
The paper's Materials & Methods lists materials provided by AminoMatrix Labs / Amelio Biomedical, and the authors acknowledge our support in the Acknowledgments. (Source: journal DOI)